Biomarkers and Why Testing is Important
Once a person has been diagnosed with lung cancer, the “standard of care” practice is to test for a panel of biomarkers that could help to determine the treatment for that particular patient. Determining which biomarker is helping to drive the cancer allows the medical care team to determine the best and most effective treatment for that specific patient.
What Do I Need to Know About Biomarker Testing?
Biomarker (or Genomic Diagnostic) testing is key in today’s world of precision medicine. Lung cancer biomarker testing is also known as tumor testing, genetic mutation testing, next generation sequencing (NGS) or genomic testing. It looks for changes in the DNA of tumor cells. These changes include mutations, additions, deletions, or rearrangements in the DNA. The “specific biomarkers” in the cells of a tumor can be used to determine the best course of treatment for a lung cancer patient.
When a lung cancer patient first receives a diagnosis of lung cancer, that is the best time to discuss biomarker testing with your doctor. No matter if you or your doctor initiate this conversation, be sure to ask about next generation sequencing (NGS; also called comprehensive biomarker testing). NGS tests for multiple biomarkers at the same time.
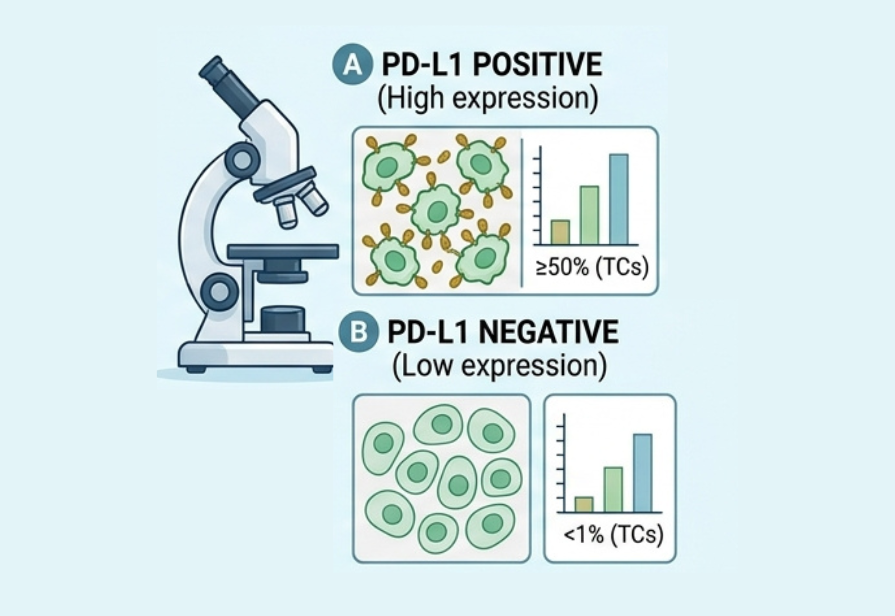
Biomarkers with FDA-approved Treatments
Questions about biomarker testing
Normally, cancer cells are collected from your tumor by a biopsy. Commonly, the tumor tissue can be removed through the skin with a needle. There are other forms of retrieving tumor tissues. Some of these are bronchoscopy, lymph node biopsy, mediastinoscopy, computed tomography (CT)–guided core needle biopsy, and via the video-assisted thoracoscopic surgery (VATS) procedure. Your healthcare team will be able to tell you which option may be right for you.
To test the tumor, there needs to be enough tissue from the biopsy sample for testing. The more tissue there is, the more likely you will be able to have all of the required or recommended tests. If you had your tumor surgically removed, there likely will be enough tissue available for testing. Your healthcare team will work with you to make sure that the lab has enough tumor cells to test.
One of the most exciting new areas of lung cancer research discovery is the rapidly changing field of using blood tests – liquid biopsies – for lung cancer biomarker testing. The first liquid biopsy for lung cancer was approved by the US Food and Drug Administration (FDA) in 2020. Some biomarkers – for example EGFR – can be identified through blood testing even before needle or other biopsy. However, if a liquid biopsy is negative, a tissue biopsy will be necessary. In most cases, liquid biopsies are not yet able to detect with certainty the presence of specific biomarkers in the bloodstream. Talk to your doctor to see if a liquid biopsy would be appropriate for you.
Comprehensive biomarker testing (next generation sequencing) is offered at many hospitals, medical centers and academic institutions. Once your oncologist orders the biomarker testing, the pathology department in the hospital is responsible for sending your tumor tissue to the appropriate testing facility.
How long it takes to get biomarker test results will depend on the amount , quality of the tumor tissue collected in the biopsy and the type of testing being requested. The time to process the biomarker testing is determined by the time it takes your local hospital to locate, prepare, and send your tumor specimen to the testing facility and the type of test requested. But, sometimes there is a need to speed up this process. For example, sometimes the treatment plan depends on the results of the biomarker testing. If this is the case, your doctor can request that the test results be processed faster.
Biomarker testing may be repeated for certain patients and for certain reasons. As an example, your doctor may order another round of biomarker testing if your lung cancer becomes resistant to a treatment that previously worked for you.
A patient being treated with a targeted therapy lung cancer drug can develop resistance to that drug after a period of time. This means that the drug stops working because the tumor cells “outsmart” the drug, therefore allowing the cells to grow. “Second and third generation treatments” are often available in clinical trials that overcome this drug resistance, and tumor cell growth can once again be contained.
If no treatable mutations are identified, a patient may be offered chemotherapy or radiation A patient can take advantage of cancer centers doing expanded testing or enroll in a clinical trial for any new targeted therapy under investigation.
If the biomarker testing (genomic diagnostic test for lung cancer) is recommended by your doctor, insurance companies will typically cover the cost. But, it is always a good idea to check with your health insurance plan to make sure, since there can be gaps in coverage.
Get up-to-date guidelines on biomarker testing
The National Comprehensive Cancer Network (NCCN) provides updated guidelines for patients and doctors to know which biomarkers to test for. These guidelines change as new knowledge is discovered by lung cancer researchers.